When molecular mass expressed in gram is called gram molecular weight. In learning chemistry, the molecular mass or weight of the compound meaning the number that indicates how heavy a molecule of the compound is compared to one-twelfth of the carbon-12 isotope. Therefore, one amu is numerically equal to its atomic weight on the carbon-12 scale, The atomic mass unit represents by the symbol amu, which means exactly one-twelfth of the mass of the carbon-12 atom. 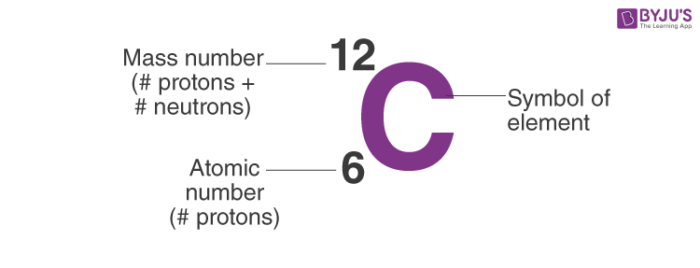
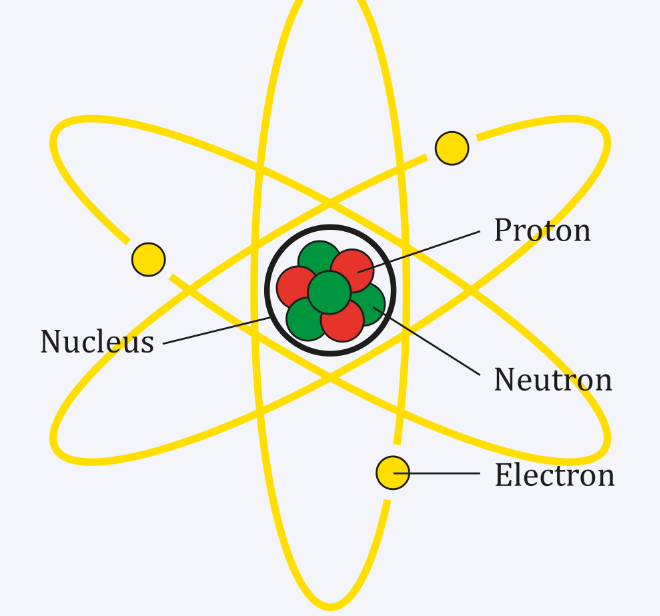
But the gram atomic weight is calculated in the gram unit. The atomic weight or mass of an element is a unitless quantity. The gram atomic weight of an element determines the mass in gram that represents the same number of atoms as twelve grams of C-12 isotope. The atomic mass of an element meaning the number that says how much heavy an atom of chemical elements is compared to one-twelfth of the carbon-12 isotope. In the chemical scale of the atomic weight or masses, the exact number 16 is taken as the reference. The exact number 16 as the relative mass of oxygen represents the new scale of atomic weights. But natural oxygen was found to be the mixture of three isotopes of mass number 16, 17, and 18 with the percentage of abundance 993575, 0.039, and 0.204 respectively. On the physical scale of atomic mass or weight, the natural oxygen atom (relative weight 16) is taken as the reference. To maintain electroneutrality, the atom must carry an equal number of extranuclear electrons. The most reasonable interpretation of the Mosely experiment, the atomic number of elements is equal to the number of protons in the atomic nucleus. A better plot was obtained when atomic weight is replaced by the order number or the serial number of the elements in the periodic table. 
A straight line plot was obtained but few elements are out of line.Mosely plotted the square root of wavelength for each element against atomic weight.Moseley experiment with over thirty metals from aluminum to gold.When a beam of high-energy electrons falls on the metal target, the atom of the metal excited and emitted radiant energy.The identification of the nuclear charge and the atomic number of an element established by electromagnetic spectrum data find from the x-ray discovered by Moseley. Rutherford model or alpha ray scattering experiments allow for the determination of the positive charge of the nucleus of an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
